



Trichomoniasis Vaginalis Culture Test
-
This test is designed for user-friendly and convenient early microscopic detection by culture confirmation of T. vaginalis.
- Highly selective, culture media detection of Trichomonas vaginalis, in a cost-effective, easy to use, broth media device
- Allows wet mount immediate observation or store/transport
- Saves time, material cost and waste, reducing exposure to collected sample
THE PROBLEM
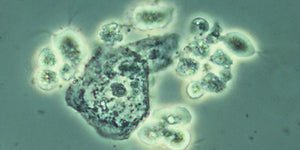
Human trichomoniasis is a sexually transmitted infection (STI) caused by the flagellated protozoan Trichomonas vaginalis (TV). It is recognized as the most prevalent non-viral sexually transmitted infections world-wide, in both males and females. TV is not a reportable disease — creating serious challenges in treating this disease. It is increasingly resistant to the most prescribed treatment, Metronidazole (MTZ). When the infection persists, the patient can develop acute urogenital inflammatory conditions, infertility, and increased vulnerability to cervical cancer and HIV.
THE SOLUTION
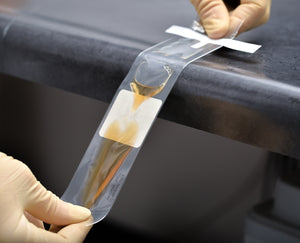
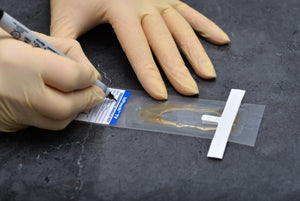
InPouch is designed for user-friendly and convenient early microscopic detection by culture confirmation of T. vaginalis. InPouch consists of a high-barrier, oxygen-resistant, plastic with two V-shaped-chambers connected by a narrow passage that, together, provide a variety of benefits — and allows users to easily inoculate a specimen, immediately observe (wet mount) the specimen, store and/or transport (optional) before transferring to the lab for incubation and recording.
- Presumptive positive results when trichomonads are observed microscopically. If negative upon initial observation, incubate at 37°C and examine every 24 hours for 5 days.
- No plating or wet mount slide preparation needed — place directly on the microscope stage for low power (100x) observation
Where is the price?
Pricing is based on shipping location and the number of units ordered. To request a price, please use the Request a Quote button below. A sales representative will respond to you quickly.
This test is designed for user-friendly and convenient early microscopic detection by culture confirmation of T. vaginalis.
- Highly selective, culture media detection of Trichomonas vaginalis, in a cost-effective, easy to use, broth media device
- Allows wet mount immediate observation or store/transport
- Saves time, material cost and waste, reducing exposure to collected sample
THE PROBLEM
Human trichomoniasis is a sexually transmitted infection (STI) caused by the flagellated protozoan Trichomonas vaginalis (TV). It is recognized as the most prevalent non-viral sexually transmitted infections world-wide, in both males and females. TV is not a reportable disease — creating serious challenges in treating this disease. It is increasingly resistant to the most prescribed treatment, Metronidazole (MTZ). When the infection persists, the patient can develop acute urogenital inflammatory conditions, infertility, and increased vulnerability to cervical cancer and HIV.
THE SOLUTION
InPouch is designed for user-friendly and convenient early microscopic detection by culture confirmation of T. vaginalis. InPouch consists of a high-barrier, oxygen-resistant, plastic with two V-shaped-chambers connected by a narrow passage that, together, provide a variety of benefits — and allows users to easily inoculate a specimen, immediately observe (wet mount) the specimen, store and/or transport (optional) before transferring to the lab for incubation and recording.
- Presumptive positive results when trichomonads are observed microscopically. If negative upon initial observation, incubate at 37°C and examine every 24 hours for 5 days.
- No plating or wet mount slide preparation needed — place directly on the microscope stage for low power (100x) observation









