




Prueba de bebés prematuros
-
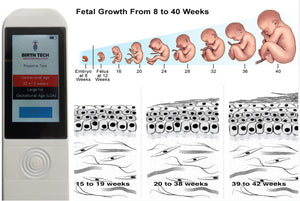
Este dispositivo fácil de operar y basado en evidencia puede evaluar la edad gestacional de un recién nacido usando luz para determinar la madurez de la piel (el dispositivo detecta a los recién nacidos prematuros analizando las propiedades fotobiológicas de la piel del recién nacido).
- La edad gestacional es el principal marcador de supervivencia neonatal
- Portátil y fácil de usar para que los no especialistas evalúen la edad gestacional y tomen decisiones clínicas sobre el transporte o la reanimación
- El dispositivo advierte sobre errores de medición
EL PROBLEMA
Según la OMS, cada año alrededor de 15 millones de bebés nacen prematuramente y 1 millón mueren debido a complicaciones de la prematuridad. Cada vez que un bebé nace demasiado pequeño, las parteras deben tomar decisiones urgentes sobre las intervenciones inmediatas y la derivación del recién nacido a un hospital. Es importante determinar de inmediato si un recién nacido es prematuro o pequeño para la edad gestacional. Sin cuidados intensivos, los recién nacidos prematuros corren el riesgo de una variedad de riesgos de mortalidad a corto plazo y de por vida. La mayoría de estas vidas podrían salvarse con la pronta identificación de la prematuridad neonatal. Sin embargo, en ausencia de un ultrasonido (a menudo demasiado costoso y no accesible) no existe un estándar de oro para la datación del embarazo.
LA SOLUCIÓNPreemie Test es el primer dispositivo médico capaz de evaluar con precisión la edad gestacional inmediatamente después del nacimiento de un bebé. La prueba del bebé prematuro cuenta con una sonda que contiene emisores y receptores de luz que se pone en contacto con el pie del recién nacido y, utilizando algoritmos matemáticos, proporciona una fecha estimada en segundos. Este dispositivo optoelectrónico no invasivo mide la madurez (grosor) de la piel a través de la retrodispersión de la luz de la piel, utilizando un diodo emisor de luz en longitudes de onda de 470 nm, 575 nm y 630 nm. Múltiples ensayos clínicos han validado la eficacia y precisión de los dispositivos Preemie Test (Brasil, Portugal, Mozambique, India y Malawi). Todos los estudios están disponibles bajo petición.
Inmediatamente después del parto, un recién nacido con edad gestacional desconocida o no confiable requiere reanimación con pasos iniciales de estabilización y ventilación con presión positiva. La diferenciación inmediata entre SGA y IUGR es fundamental, al igual que determinar el nivel de prematuridad (en semanas) con certeza.
La batería de este dispositivo tiene una duración de tres años, lo que permite cientos o miles de mediciones. Según la agencia reguladora de salud de Brasil (ANVISA), este dispositivo médico está categorizado como Clase II de seguridad: no invasivo y de riesgo medio.
¿Dónde está el precio?
El precio se basa en la ubicación de envío y el número de unidades pedidas. Para solicitar un precio, utilice el botón Solicitar una cotización a continuación. Un representante de ventas le responderá rápidamente.
Este dispositivo fácil de operar y basado en evidencia puede evaluar la edad gestacional de un recién nacido usando luz para determinar la madurez de la piel (el dispositivo detecta a los recién nacidos prematuros analizando las propiedades fotobiológicas de la piel del recién nacido).
- La edad gestacional es el principal marcador de supervivencia neonatal
- Portátil y fácil de usar para que los no especialistas evalúen la edad gestacional y tomen decisiones clínicas sobre el transporte o la reanimación
- El dispositivo advierte sobre errores de medición
EL PROBLEMA
Según la OMS, cada año alrededor de 15 millones de bebés nacen prematuramente y 1 millón mueren debido a complicaciones de la prematuridad. Cada vez que un bebé nace demasiado pequeño, las parteras deben tomar decisiones urgentes sobre las intervenciones inmediatas y la derivación del recién nacido a un hospital. Es importante determinar de inmediato si un recién nacido es prematuro o pequeño para la edad gestacional. Sin cuidados intensivos, los recién nacidos prematuros corren el riesgo de una variedad de riesgos de mortalidad a corto plazo y de por vida. La mayoría de estas vidas podrían salvarse con la pronta identificación de la prematuridad neonatal. Sin embargo, en ausencia de un ultrasonido (a menudo demasiado costoso y no accesible) no existe un estándar de oro para la datación del embarazo.
LA SOLUCIÓNPreemie Test es el primer dispositivo médico capaz de evaluar con precisión la edad gestacional inmediatamente después del nacimiento de un bebé. La prueba del bebé prematuro cuenta con una sonda que contiene emisores y receptores de luz que se pone en contacto con el pie del recién nacido y, utilizando algoritmos matemáticos, proporciona una fecha estimada en segundos. Este dispositivo optoelectrónico no invasivo mide la madurez (grosor) de la piel a través de la retrodispersión de la luz de la piel, utilizando un diodo emisor de luz en longitudes de onda de 470 nm, 575 nm y 630 nm. Múltiples ensayos clínicos han validado la eficacia y precisión de los dispositivos Preemie Test (Brasil, Portugal, Mozambique, India y Malawi). Todos los estudios están disponibles bajo petición.
Inmediatamente después del parto, un recién nacido con edad gestacional desconocida o no confiable requiere reanimación con pasos iniciales de estabilización y ventilación con presión positiva. La diferenciación inmediata entre SGA y IUGR es fundamental, al igual que determinar el nivel de prematuridad (en semanas) con certeza.
La batería de este dispositivo tiene una duración de tres años, lo que permite cientos o miles de mediciones. Según la agencia reguladora de salud de Brasil (ANVISA), este dispositivo médico está categorizado como Clase II de seguridad: no invasivo y de riesgo medio.








